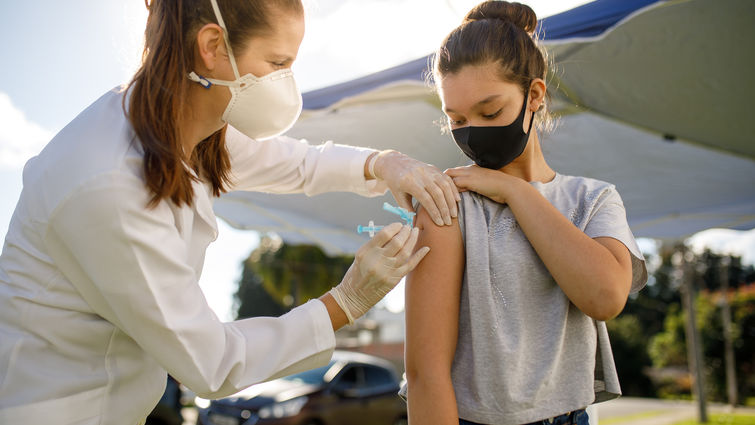
The Centers for Disease Control and Prevention have announced their emergency use authorization for Pfizer-BioNTech’s COVID-19 vaccine for use in adolescents ages 12-15 years old, a pivotal step in increasing eligibility for the vaccine in the United States. This approval comes after the Food and Drug Administration’s emergency use sign-off on Monday.
Alexandra Clark, MD, division chief of general pediatrics and pediatric medicine for Loma Linda University Children’s Health, answers questions about vaccine safety in adolescents, side effects, and more.
Adolescents are generally young and healthy — why do they need to be vaccinated against COVID-19?
While it is true that the youth of America have suffered less with the original COVID-19 disease than the elderly, they have been the biggest bearers of the burden of multi-system inflammatory syndrome in children (MIS-C), which is a disease process that occurs one to three weeks after the original infection. MIS-C has had severe consequences for children, including long-term cardiac consequences, as well as other major organ system damage. Doctors can't predict which child is going to suffer this inflammatory response syndrome and which child will not, so any child could develop these long-term, serious side effects from COVID-19, even if the child was asymptomatic. Additionally, teens are as likely to spread the virus at the same rate as adults. If they do get the infection and bring it home to an immunocompromised family member or elderly parent or grandparent, they're more likely to cause a home infection having gone out and about in the community and acquiring the disease, even if they're not very sick themselves. Vaccinating that part of the population is critical to keeping our homes safe and protecting those more vulnerable in the community. In addition, the American Academy of Pediatrics just released data to show that we are seeing a rising percentage of new cases being children, representing over 22% of new cases.
The vaccine is so new — is it safe for kids?
This Pfizer vaccine to date is shown to be one of the safest vaccines ever brought to market with millions of people worldwide having been vaccinated. We have a lot of good safety data about how this vaccine is working and how effective it is against protecting from the original type of COVID-19 as well as the variant strains. I know it’s hard for parents and families to not be scared of something they don’t know. I want to remind people that while Pfizer’s MRNA vaccine is cutting-edge technology, it’s not new technology; rather, it’s being used in a new way to bring this vaccine to market. The MRNA vaccine is not utilizing any real virus. Because it’s not injecting anything into the host DNA, there are no long-term repercussions of having this sort of blueprint put in your body. The cell that takes the MRNA vaccine will use it to create the antibody response. Then, as that cell goes through its natural cycle, it will die and we’re left with the immune protections but without having anything adjusted or altered in our own DNA or cells.
How effective is the vaccine in preventing COVID-19 in this age group?
In the clinical setting for 12-15-year-olds, the vaccine was shown to be 100% effective in preventing COVID-19. For this Pfizer vaccine, the dosage for this age group will be the same with the two shots.
What are the side effects of the vaccine in adolescents?
Side effects were equivalent to what adults experienced in the vaccine trial and now in the general population — largely short-lived pain at the injection site, fatigue, muscle aches, body aches, and headache. It’s one of the safest vaccines to have been invented.
What steps were taken by the FDA and CDC before the vaccine was approved for emergency use for adolescents?
For vaccines to be authorized for any age range, there had to be three phases of a clinical trial. After the trial was done, which was with 100% volunteer enrollment across multi-demographic regions across the U.S., the data was submitted to the FDA for review. The FDA reviewed the trial, looking at the rigor of the data and ensured that what was turned in was accurate. They then made a recommendation to the CDC. The CDC met to discuss effectiveness, safety, and immune response before voting on whether or not to recommend the vaccine for adolescents.
If my child already had COVID-19 previously, do they still need to be vaccinated?
Yes, getting the vaccine has been proven to provide additional, significant protection against reacquiring COVID-19. Data is showing that teens’ immune systems are well primed to create a good response to the vaccine and are likely to be well protected as a result of getting the vaccine. Just having the novel infection itself, particularly if the child was actually asymptomatic, is less likely to provide you long-term immune protection.
What if my adolescent doesn’t want the vaccine?
This is not a mandatory vaccine at this point; however, there are some school districts, higher education institutions included, that are considering whether to require the vaccine for their students to be on campus in the fall.
If someone doesn’t want the vaccine, I recommend they have a transparent conversation with their physician about their concerns related to the vaccine. It’s everyone’s right to have a good, informed decision — I would just ask parents and their kids to have that discussion with their healthcare provider to talk about their concerns before they make a choice.
Loma Linda University Health is now offering vaccinations for adolescents ages 12-15. Learn more about receiving the COVID-19 vaccination by visiting our website.